Have you ever wondered how your favorite spirits or essential oils are crafted? Fractional distillation examples reveal the fascinating science behind separating mixtures into their pure components. This method isn’t just for lab coats and beakers; it’s widely used in industries from petrochemicals to food production, showcasing its versatility and importance.
In this article, you’ll explore various examples of fractional distillation, highlighting processes that turn crude oil into gasoline and separate complex mixtures in laboratories. Each example illustrates how this technique optimizes efficiency while ensuring high purity levels. By the end, you’ll gain a deeper understanding of how fractional distillation shapes everyday products and industries. Are you ready to dive into the world of separation science?
Overview Of Fractional Distillation
Fractional distillation plays a crucial role in various industries by separating mixtures into their individual components. You might encounter it in everyday products without realizing its significance. Here are some key examples that illustrate how fractional distillation operates:
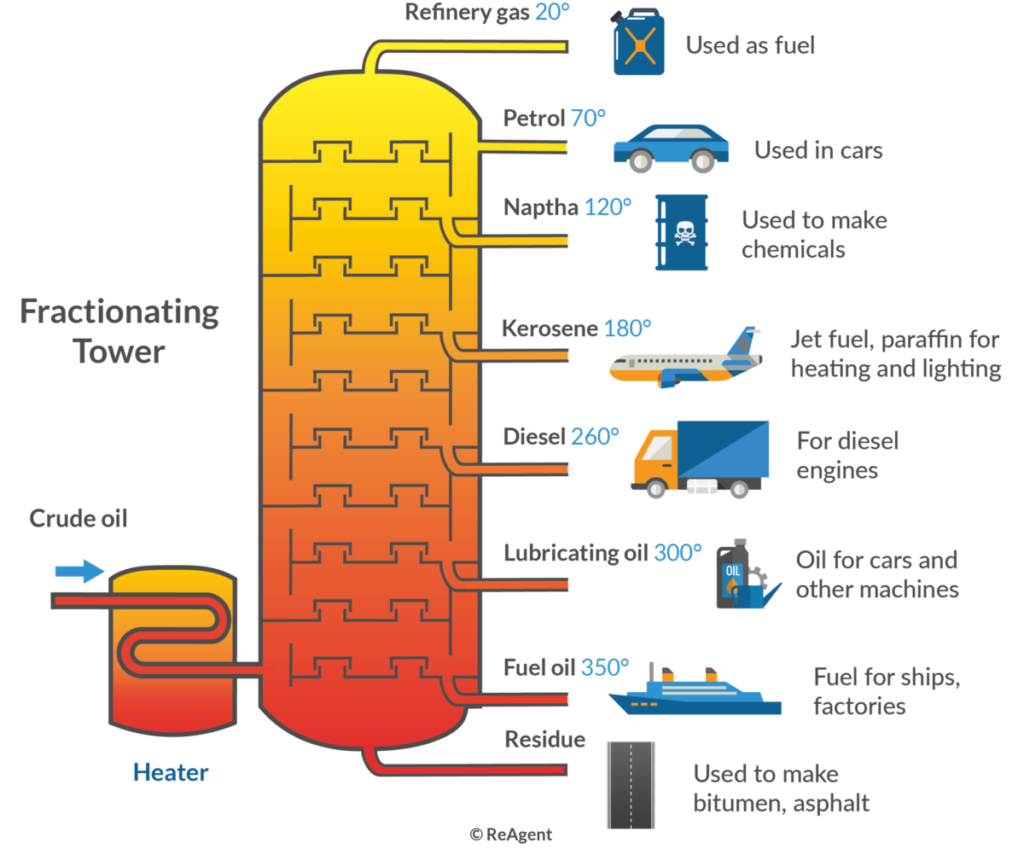
- Petroleum Refining: In this process, crude oil is heated and separated into different hydrocarbons like gasoline, diesel, and kerosene through fractional distillation columns.
- Alcohol Production: Distilleries use this method to purify ethanol from fermented mixtures. The higher boiling point of water allows for the separation of alcohol at lower temperatures.
- Chemical Manufacturing: Various chemicals such as solvents and acids undergo fractional distillation to achieve high purity levels essential for industrial applications.
- Natural Gas Processing: This technique separates methane from heavier hydrocarbons and impurities, optimizing gas quality for distribution.
These examples demonstrate how fractional distillation enhances product efficiency and purity across multiple sectors. Each application relies on precise temperature control to ensure accurate separation based on boiling points.
Common Examples Of Fractional Distillation
Fractional distillation plays a vital role in multiple industries. Here are some common examples that illustrate its applications.
Example 1: Crude Oil Refining
In crude oil refining, fractional distillation separates various hydrocarbons based on their boiling points. The process begins with heating crude oil in a distillation column. As the temperature rises, different fractions vaporize and ascend through trays at varying heights. Light fractions like gasoline condense at lower levels, while heavier ones like diesel form higher up. This method is crucial for producing fuels and other products from crude oil efficiently.
Example 2: Separation Of Ethanol And Water
The separation of ethanol and water demonstrates fractional distillation’s effectiveness in alcohol production. During fermentation, a mixture forms containing around 10% to 15% ethanol by volume along with water. By applying heat, ethanol evaporates at about 78°C, while water remains liquid until it reaches 100°C. Collecting the vapor allows you to achieve high-purity ethanol suitable for beverages or industrial use.
Example 3: Purification Of Acetic Acid
Acetic acid purification also relies on fractional distillation. Typically produced through fermentation or chemical synthesis, acetic acid can contain impurities that affect its quality. By heating the mixture in a fractionating column, you can separate acetic acid from other components based on boiling point differences—acetic acid boils at approximately 118°C while many impurities boil at different temperatures. This results in high-purity acetic acid, essential for food preservation and industrial applications.
Applications Of Fractional Distillation
Fractional distillation plays a crucial role in various industries. Here are some key applications and examples:
- Petroleum Refining: In this process, crude oil undergoes heating in a distillation column. The method separates hydrocarbons into lighter fractions like gasoline, kerosene, and diesel. Each fraction collects at different levels due to its boiling point.
- Alcohol Production: Ethanol purification occurs through fractional distillation of fermented mixtures. This technique effectively removes water and other impurities, yielding high-purity ethanol for beverages or industrial uses.
- Chemical Manufacturing: Factories use fractional distillation to produce solvents and acids with high purity levels. For instance, separating isopropanol from impurities ensures that the final product meets industry standards.
- Natural Gas Processing: This application optimizes gas quality by separating methane from heavier hydrocarbons like ethane and propane. The separation enhances energy efficiency in natural gas usage.
These examples illustrate how fractional distillation contributes significantly to product efficiency and purity across multiple sectors.
Benefits And Limitations Of Fractional Distillation
Fractional distillation offers several advantages in various industries. For instance, it allows for the separation of mixtures into their pure components efficiently. This method is particularly useful in petroleum refining, where it helps produce high-quality fuels like gasoline and diesel. Furthermore, its precision in temperature control ensures accurate separation based on boiling points, enhancing product quality across sectors.
However, fractional distillation also has limitations. The process can be energy-intensive, leading to higher operational costs in some cases. Additionally, it may not effectively separate components with close boiling points, resulting in less purity for certain products. Moreover, the installation and maintenance of distillation columns require significant investment.
Consider these specific examples:
- In crude oil refining, lighter fractions are separated from heavier ones.
- Alcohol production involves separating ethanol from water through careful temperature adjustments.
- Acetic acid purification leverages boiling point differences to isolate high-purity acetic acid.
These examples illustrate how fractional distillation plays a vital role while also highlighting its challenges within industrial applications.