Have you ever wondered how a simple chemical reaction can produce heat? Exothermic reactions are fascinating processes where energy is released, often in the form of warmth. These reactions play a crucial role in everyday life, from combustion to biological processes.
Overview Of Exothermic Reactions
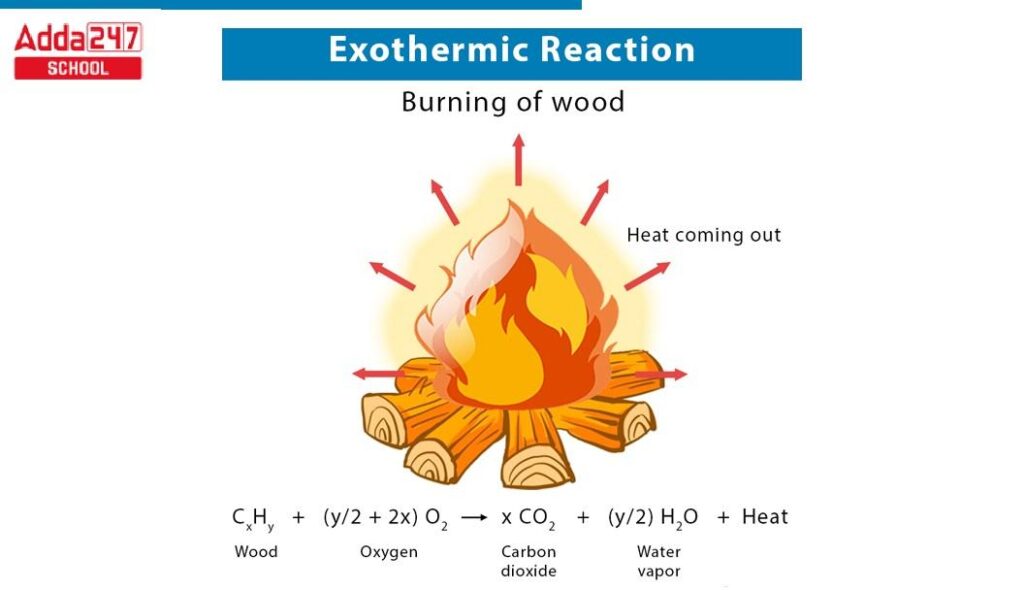
Exothermic reactions are chemical processes that release energy, primarily in the form of heat. These reactions play a crucial role in everyday life and various natural processes. Here are some common examples:
- Combustion: When fuels like gasoline or wood burn, they undergo combustion, releasing significant amounts of heat and light.
- Respiration: In your body, cellular respiration converts glucose into energy. This process releases heat as a byproduct.
- Thermite Reaction: Mixing aluminum powder with iron oxide results in a thermite reaction. This exothermic process generates intense heat and produces molten iron.
- Neutralization: When an acid reacts with a base, such as hydrochloric acid with sodium hydroxide, it forms water and salt while releasing heat.
- Hydration of Cement: The curing process of cement involves exothermic reactions that generate warmth as the material hardens.
Understanding these examples helps illustrate how exothermic reactions are integral to both industrial applications and biological functions.
Common Exothermic Reaction Examples
Exothermic reactions are prevalent in daily life and industrial processes. Here are some common examples showcasing their significance.
Combustion Reactions
Combustion reactions release a significant amount of energy in the form of heat and light. Common fuels involved include:
- Gasoline: Used in vehicles, it combusts with oxygen, producing carbon dioxide and water.
- Wood: Burning wood for heat or cooking releases energy through combustion.
- Natural Gas: Often used for heating, natural gas combustion emits carbon dioxide and water vapor.
Respiration Processes
Cellular respiration is an essential exothermic reaction that occurs in living organisms. This process involves:
- Glucose Breakdown: Cells convert glucose into energy, releasing carbon dioxide and water as byproducts.
- Aerobic Respiration: In the presence of oxygen, organisms extract energy from food effectively.
- Hydrochloric Acid and Sodium Hydroxide: Mixing these produces sodium chloride (table salt) and water with a noticeable temperature increase.
- Acetic Acid and Sodium Bicarbonate: This reaction also generates heat along with carbon dioxide gas bubbles.
Applications Of Exothermic Reactions
Exothermic reactions play crucial roles in various applications across industries and everyday life. These reactions not only produce energy but also contribute to essential processes.
Energy Production
Energy production relies heavily on exothermic reactions. Combustion of fossil fuels like coal, oil, and natural gas generates heat energy that powers electricity plants. This process transforms chemical energy into thermal energy, which then creates steam to drive turbines for electricity generation. Also, biomass burning, such as wood or agricultural waste, serves as a renewable source of energy while releasing heat during combustion.
Industrial Processes
Various industrial processes utilize exothermic reactions for efficiency. Cement hydration exemplifies this; when water mixes with cement powder, it releases significant heat, aiding the curing process and enhancing structural integrity. Similarly, thermite reactions are used in metal welding and cutting; they involve aluminum powder reacting with iron oxide to produce molten iron along with intense heat for joining metals effectively. Additionally, many manufacturing processes involving acids and bases leverage neutralization reactions to generate heat necessary for certain chemical transformations.
Safety Considerations
Exothermic reactions can release significant amounts of heat and energy, requiring careful handling. Always wear appropriate personal protective equipment (PPE) like gloves and goggles when conducting experiments.
Consider these safety measures:
- Ventilation: Ensure adequate ventilation in the workspace to avoid the buildup of harmful gases.
- Storage: Store chemicals properly, following guidelines for flammable and reactive materials.
- Emergency Procedures: Familiarize yourself with emergency procedures, including first aid for burns or chemical exposure.
You might also want to keep a fire extinguisher nearby when performing exothermic reactions. Understanding these safety precautions helps prevent accidents and injuries while working with potentially hazardous materials.