Imagine stepping outside and seeing the world around you slowly fading away. That’s the reality of acid rain, a phenomenon that affects not just our environment but also our health and infrastructure. What causes this acidic downfall, and why should you care?
In this article, you’ll discover how acid rain forms from pollutants like sulfur dioxide and nitrogen oxides. You’ll learn about its devastating impact on ecosystems, buildings, and even human health. From damaged forests to corroded monuments, the effects of acid rain are far-reaching and often underestimated. Are we doing enough to combat this growing threat?
Understanding Acid Rain
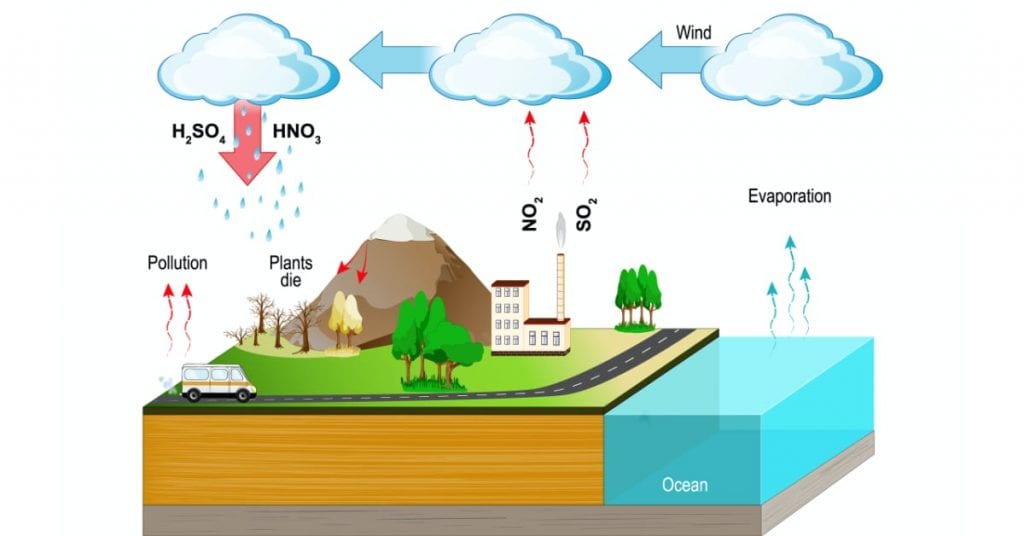
Acid rain refers to any form of precipitation that has a lower pH than normal, typically below 5.6. This phenomenon results from air pollutants mixing with moisture in the atmosphere, leading to environmental concerns.
Definition of Acid Rain
Acid rain is not just limited to rain; it includes snow, fog, and dust that carry acidic components. When these forms of precipitation fall to the ground, they can cause harm to the environment. The term encompasses various acidic conditions arising from human activities and natural sources.
Causes of Acid Rain
Pollutants such as sulfur dioxide (SO2) and nitrogen oxides (NOx) are the primary culprits behind acid rain. These compounds often originate from industrial processes, vehicle emissions, and power plants. Once released into the atmosphere:
- Sulfur dioxide transforms into sulfuric acid when combined with water vapor.
- Nitrogen oxides convert into nitric acid under similar conditions.
Both acids then mix with precipitation, creating an acidic solution that falls back to Earth.
Environmental Impact of Acid Rain
Acid rain significantly affects ecosystems, water quality, and infrastructure. Its consequences extend beyond immediate damage, impacting long-term environmental health.
Effects on Forests and Vegetation
Acid rain harms forests by leaching essential nutrients from the soil. For example:
- Nutrient depletion: Important elements like calcium and magnesium decrease, affecting tree growth.
- Tree health: Species such as maple and spruce show increased susceptibility to diseases due to weakened conditions.
- Biodiversity loss: Sensitive plant species decline because they can’t adapt to acidic environments.
You might notice that some forests appear stunted or less vibrant in areas with high acid rain levels.
Impact on Water Bodies
Water bodies also suffer from acid rain’s effects. Specifically:
- pH imbalance: Lakes and streams experience significant drops in pH levels, harming aquatic life.
- Fish populations: Species like trout are particularly vulnerable; even slight acidity can lead to reproductive failures.
- Ecosystem disruption: Changes in fish populations affect food webs, leading to broader ecological impacts.
The next time you visit a lake or river, consider the delicate balance of its ecosystem—acid rain poses a serious threat to it.
Human Health Implications
Acid rain poses significant risks to human health, impacting respiratory systems and causing skin irritations. Understanding these implications is crucial for addressing the consequences of this environmental issue.
Respiratory Issues
Exposure to acid rain can lead to Respiratory Issues. Pollutants like sulfur dioxide and nitrogen oxides contribute to air quality deterioration. These substances mix with moisture, forming acidic compounds that can cause or exacerbate conditions such as:
- Asthma: Increased attacks from inhaling polluted air.
- Chronic Bronchitis: Symptoms worsen due to higher levels of acidity in the atmosphere.
- Respiratory Infections: Higher susceptibility when breathing contaminated air.
You might notice more people experiencing difficulty breathing in areas affected by acid rain.
Skin Irritations
Acid rain doesn’t just impact the lungs; it also causes Skin Irritations. When rain falls in an acidic state, it can affect your skin upon contact, leading to problems like:
- Rashes: Irritation that may appear after exposure.
- Dryness: Skin losing moisture and becoming rough or flaky.
- Allergic Reactions: Heightened sensitivity may trigger reactions in some individuals.
Have you ever felt discomfort after being caught in a heavy downpour? Acidic precipitation could be a contributing factor.
Mitigation Strategies
Mitigation strategies play a crucial role in reducing the impacts of acid rain. These approaches focus on regulatory measures and technological innovations that address pollution sources.
Regulatory Measures
Regulatory measures are vital for controlling emissions that contribute to acid rain. Governments implement policies like:
- Clean Air Act: This U.S. law regulates air pollutants, targeting sulfur dioxide and nitrogen oxides from industrial and vehicular sources.
- Emission Trading Systems: Programs allow companies to buy and sell emission allowances, creating financial incentives to reduce pollutants.
- Fuel Standards: Implementing stricter fuel standards reduces the sulfur content in fuels used by vehicles and power plants.
These regulations aim to lower harmful emissions, thereby decreasing acid rain occurrences.
Technological Innovations
Technological innovations offer effective solutions for mitigating acid rain effects. Key advancements include:
- Scrubbers: Installed in power plants, scrubbers remove sulfur dioxide from flue gases before they enter the atmosphere.
- Catalytic Converters: Found in vehicles, these devices convert nitrogen oxides into less harmful substances before release.
- Renewable Energy Sources: Utilizing wind, solar, or hydroelectric power minimizes reliance on fossil fuels, significantly cutting emissions.
Investing in these technologies not only curtails acid rain risks but also promotes cleaner air quality overall.